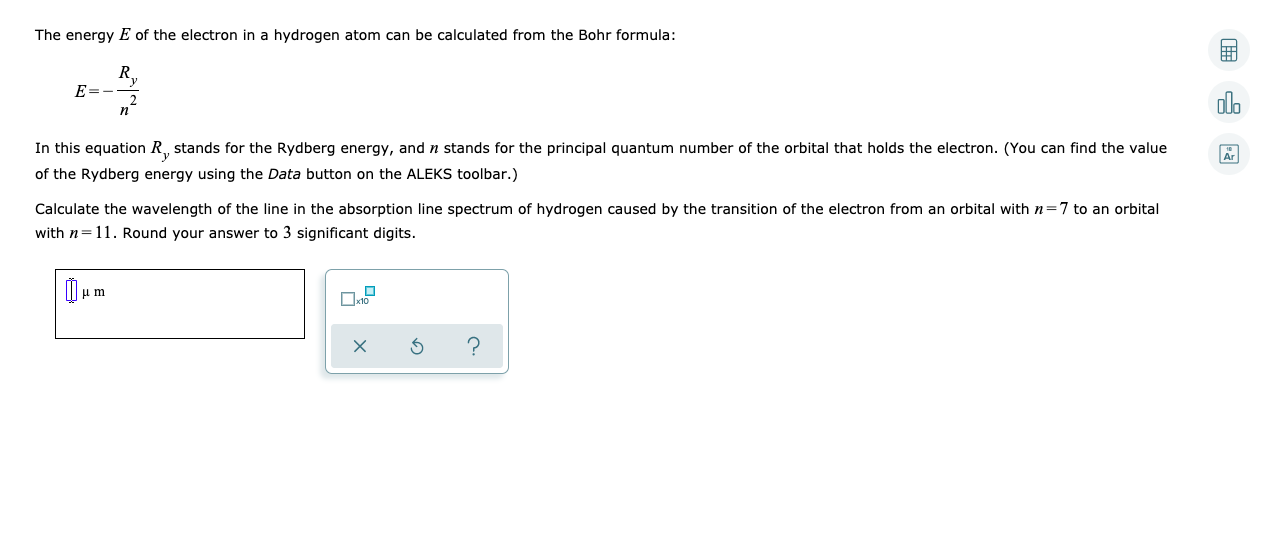
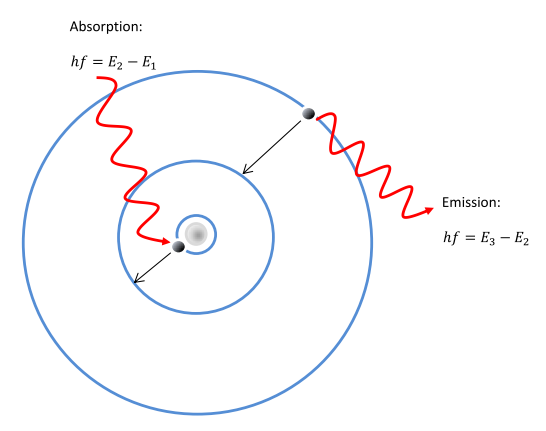
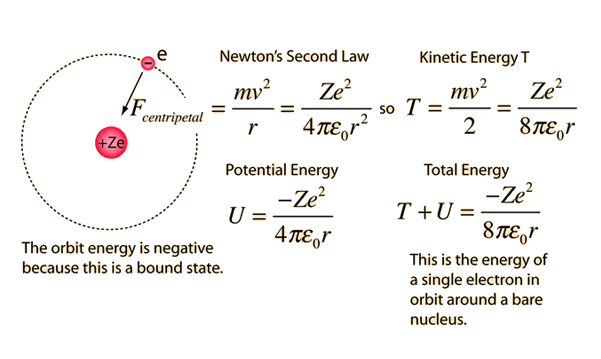
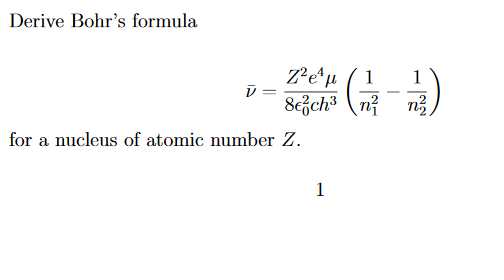Would the Bohr formula for the H-atom remain unchanged if proton had a charge (+4//3) e and electron a charge (-3//4) e, where e = 1.6 xx 10^(-19) C. Given reasons for you answer.

Using Bohr's formula for energy quantization, the ionisation potential of the ground state of Li^++ atoms is?
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

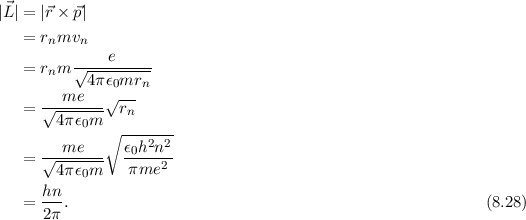





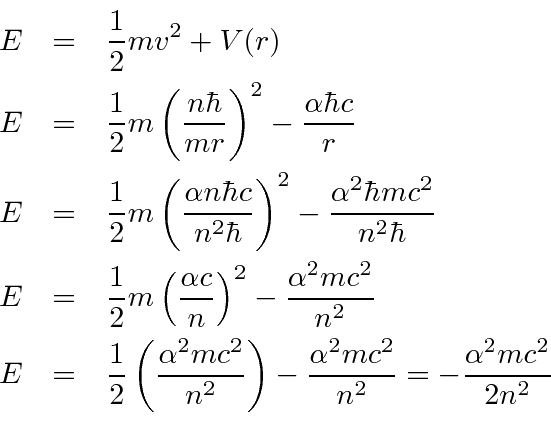




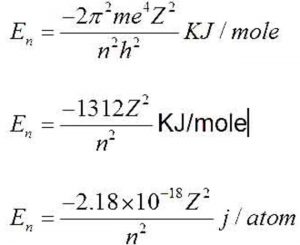

![Bohrsches Atommodell • Erklärung, Postulate, Formulierung · [mit Video] Bohrsches Atommodell • Erklärung, Postulate, Formulierung · [mit Video]](https://d3f6gjnauy613m.cloudfront.net/system/production/videos/001/833/2d5f6fe9ffd6a0af5733f881b21a0a9d4ccb0609/Bohrsches_Atommodell_Thumbnail.png?1628090256)