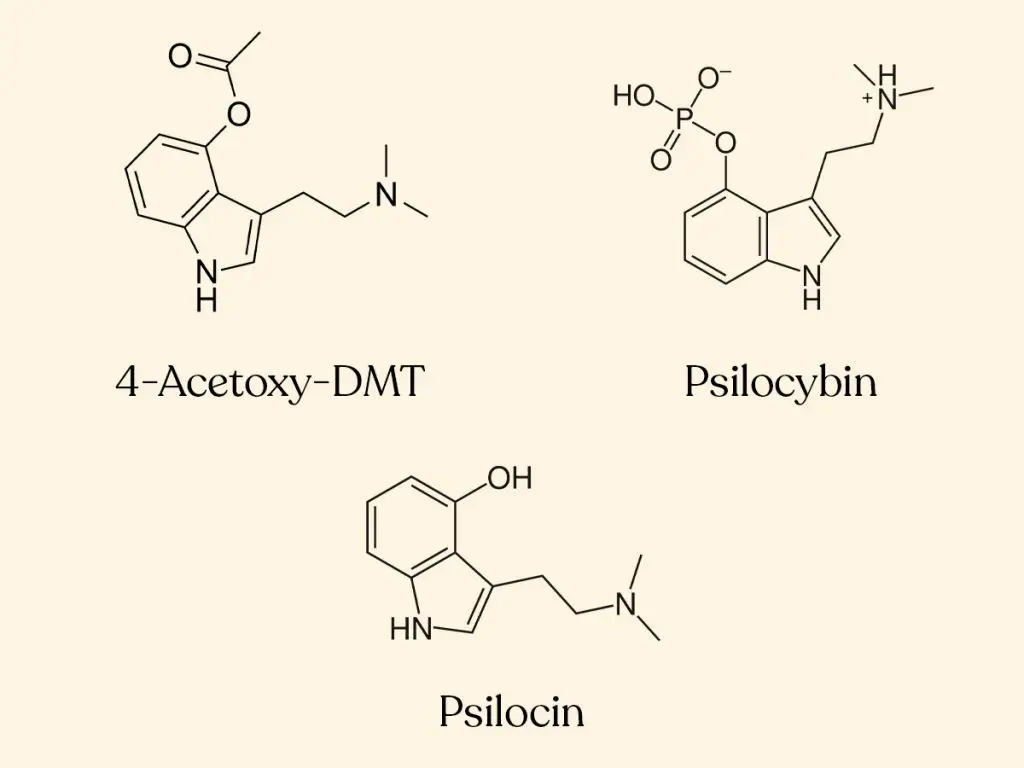
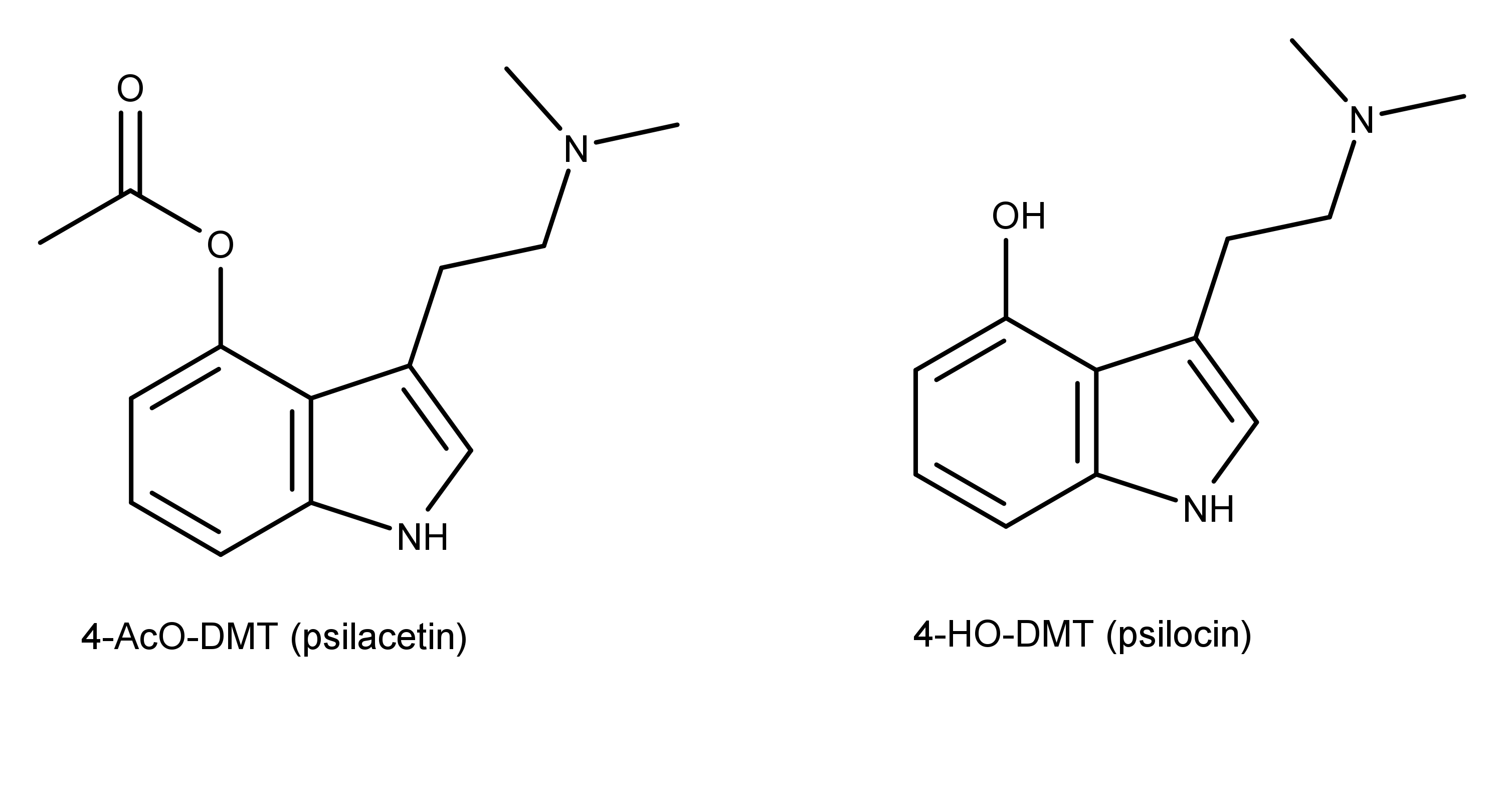
![4-AcO-MET Fumarate | 3-[2-(Ethylmethylamino)ethyl]-1H-indol-4-ol 4-Acetate Fumarate; 4-Acetoxy-N-methyl-N-ethyltryptamine Fumarate | C₁₉H₂₄N₂O₆ | TRC 4-AcO-MET Fumarate | 3-[2-(Ethylmethylamino)ethyl]-1H-indol-4-ol 4-Acetate Fumarate; 4-Acetoxy-N-methyl-N-ethyltryptamine Fumarate | C₁₉H₂₄N₂O₆ | TRC](https://www.trc-canada.com/prod-img/A190275.png)
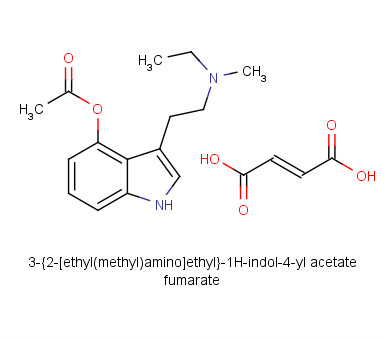
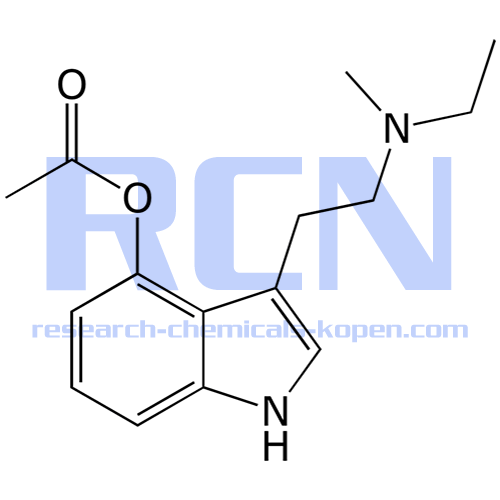
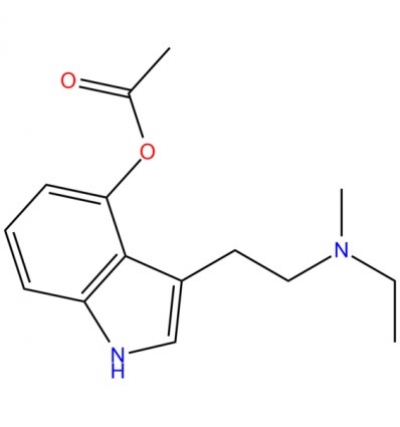
4-AcO-MET Fumarate | 3-[2-(Ethylmethylamino)ethyl]-1H-indol-4-ol 4-Acetate Fumarate; 4-Acetoxy-N-methyl-N-ethyltryptamine Fumarate | C₁₉H₂₄N₂O₆ | TRC

4-Acetoxy-N-methyl-N-ethyltryptamine Fumarate (4-AcO-MET Fumarate; 4-Acetoxy -MET Fumarate) 1.0 mg/ml in Dimethyl Sulfoxide (as free base)
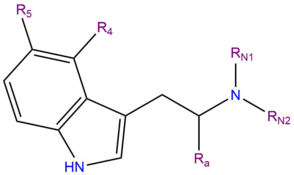
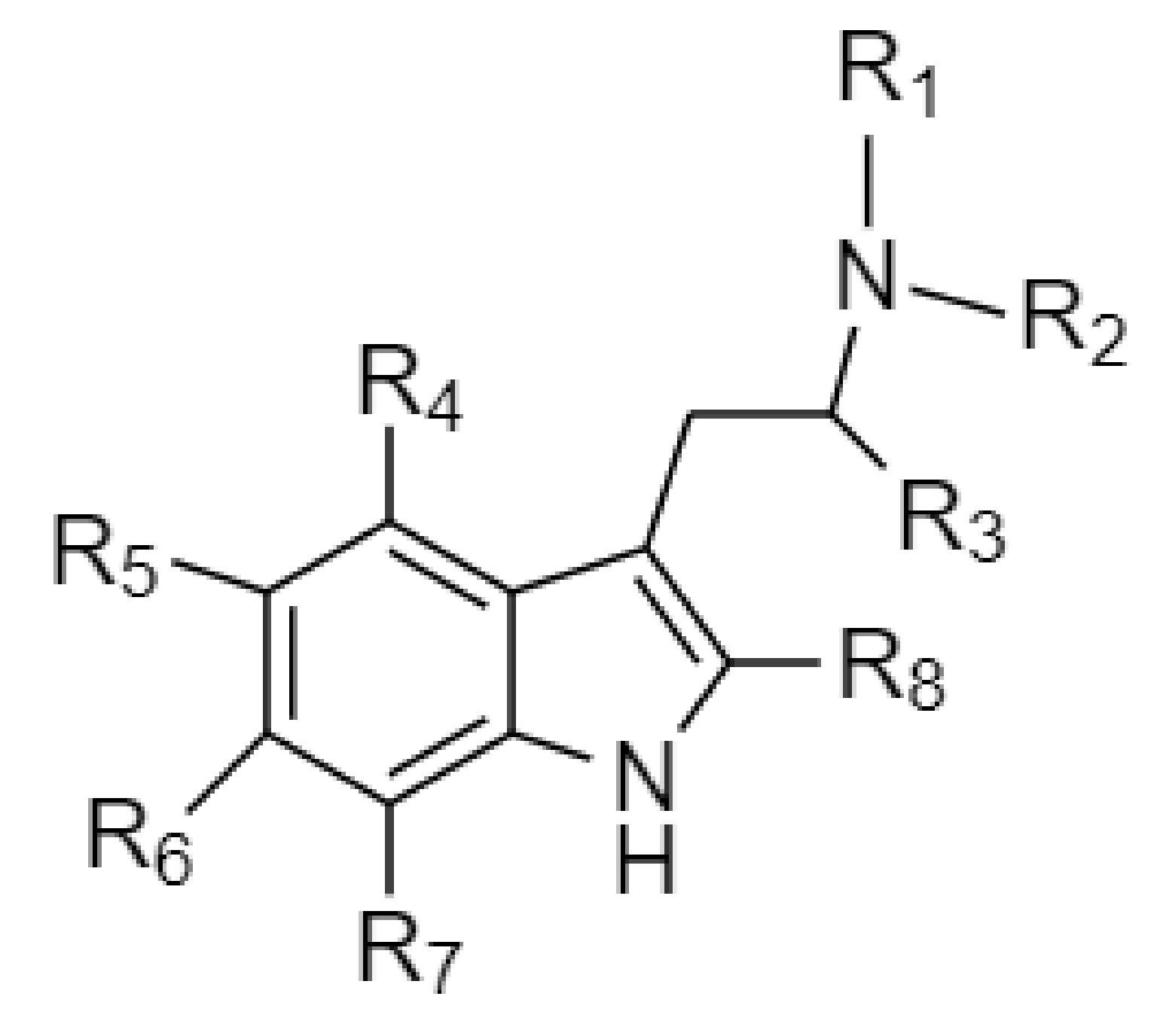
Psilacetin derivatives: fumarate salts of the meth­yl–ethyl, meth­yl–allyl and diallyl var